Progetto finanziato in Horizon 2020
Fast track to Innovation
Acronimo: GLAUrious
Link: www.glaurious.eu
Titolo: External Automatic Glaucoma Laser (EAGLE) for the first-line glaucoma treatment: Commercial prototype development and validation
Bando: H2020-FTIPilot-2015-1
Schema di finanziamento: Progetto di innovazione (IA)
Responsabile scientifico: Carlo Traverso
Dipartimento: DINOGMI
Ruolo: Partecipante
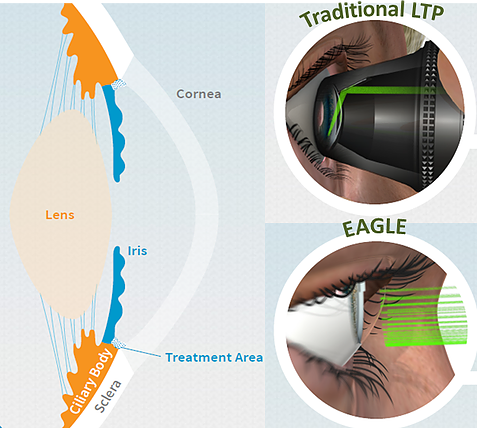
The GLAUrious project is clinically testing and validating the novel External Automatic Glaucoma Laser (EAGLE) device developed and patented by BELKIN Laser Ltd with the goal of providing accessible first-line treatment for glaucoma. About 64 million people worldwide suffer from glaucoma Which is a chronic disease causing progressive optic nerve degeneration, is the leading cause of irreversible blindness globally. Although incurable, disease progression can be arrested or delayed by reducing the pressure inside the eye, thus preventing visual incapacitation and blindness.
Current first-line treatment requires life-long daily eye drops, a strict regimen causing 50% patient dropout after 1 year. Laser treatment for glaucoma (Laser Trabeculoplasty, LTP) has been in use for many years. Its effect lasts for 2−5 years and is repeatable. This traditional 10-minute procedure, involving contact with the patient’s eye, requires the expertise of glaucoma specialists. Consequently, costs are high and accessibility to the general population is low. The novel EAGLE procedure is automated, non-invasive, non-contact, painless and user friendly—1 second of treatment by one press of a button. This revolutionary cost-effective device could be safely used by all the world’s 212,000 ophthalmologists, thus becoming a primary treatment for glaucoma, overcoming serious problems of eye drop non-compliance and non-availability, thus effectively preventing disease deterioration. GLAUrious project aims to facilitate commercialization of the EAGLE device by securing clinical prototype validation, streamlining industrial production, optimizing component costs, disseminating to key opinion leaders and distributors, and refining commercial strategy. Validation of EAGLE will allow its commercialization within 3 years of project initiation, enabling penetration of the ophthalmic market and boosting of the industrial consortium partners (BELKIN Laser, Frankfurt Laser Company and Medical Consulting and Investment Company). The multidisciplinary GLAUrious Consortium, with its five partners from four member states (Belgium, Germany, Italy, UK) and one associated state (Israel), incorporates three SMEs and two academic hospitals.